Under the old CMS 14-Day Rule, reference and independent laboratories were barred from billing Medicare directly for molecular pathology tests ordered less than 14 days after an outpatient was discharged from the hospital.
Besides the obvious administrative challenges this created for both labs and hospitals, there was also clinical concern voiced over potential delays in patient testing, access to test results, and implementation of treatment plans as the 14-day timeframe is accommodated. That changed recently when the agency published its Outpatient Prospective Payment System Final Rule for 2018.
The new carve-out exceptions to the 14-day rule allows labs to bill Medicare directly under the Clinical Laboratory Fee Schedule for molecular pathology tests and advanced diagnostic laboratory tests that are excluded from OPPS packaging rules and ordered fewer than 14 days after a patient’s outpatient hospital discharge. If these criteria are met, the DOS for the excepted tests would be the date of testing rather than the date of specimen collection. The new rule went into effect with Jan. 1, 2018, date of service. This does not change the inpatient 14-day rule.
The DOS is now the date the test was performed instead of the date the specimen was obtained if these conditions are met:
- The physician orders the test following the date of a hospital outpatient’s discharge from the hospital outpatient department.
- The specimen was collected from a hospital outpatient during an outpatient encounter.
- It would be medically inappropriate to have collected the sample from the hospital outpatient other than during the hospital outpatient encounter.
- The results of the test do not guide treatment provided during the hospital outpatient encounter.
- The test was reasonable and medically necessary for the treatment of an illness.
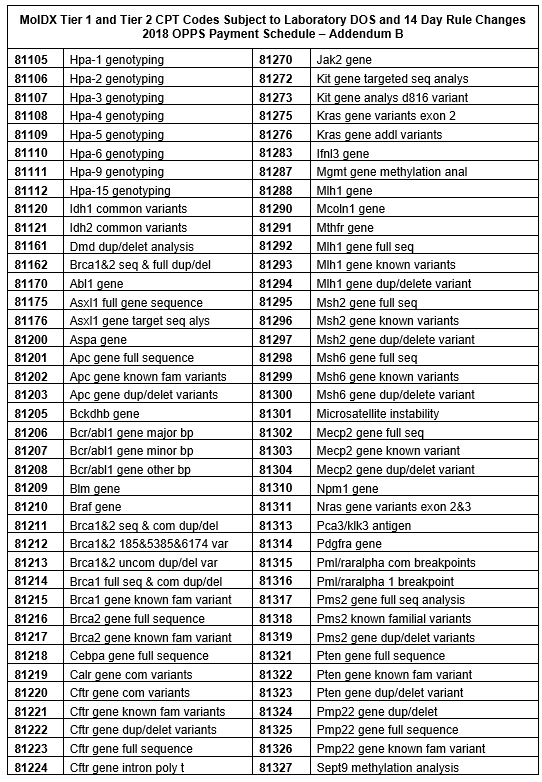
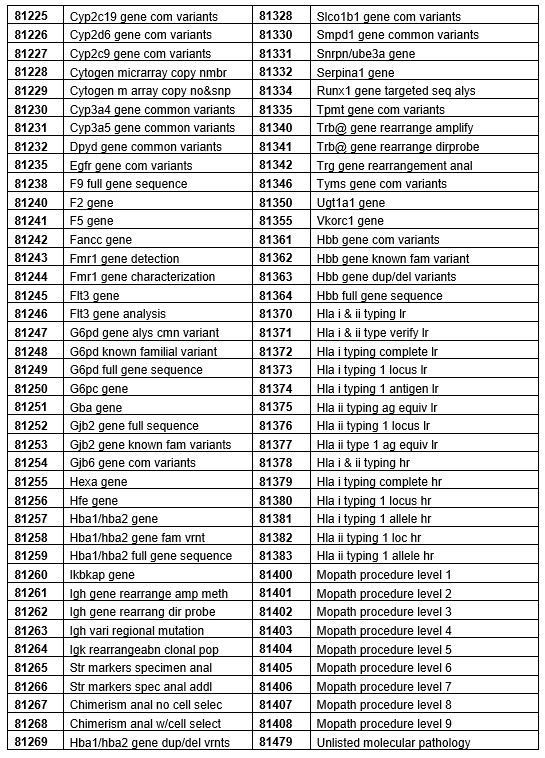
Below is a full list of affected codes.
Interested in learning more about the benefits of auditing or overall revenue cycle management? Contact us today for a free consultation.