Did you know labs can administer the COVID-19 vaccine? They absolutely CAN! I have a clinical medical lab client based in California who we recently assisted in completing this relatively simple process!
Initial steps and coding
The first step is to register as a “Mass Immunizer” with CMS. Once accepted, this status allowed them to be able to administer the vaccine. During the height of the COVID pandemic, they administered a higher volume of vaccines than they did COVID-19 testing. I was fascinated to see their innovative contributions to their community.
It’s also been interesting to help them set up their billing for vaccine administration. The laboratory cannot get paid for the vaccine itself because it has been issued to them for free from the government. However, there is reimbursement for the administering of the vaccine, as illustrated in the table below:
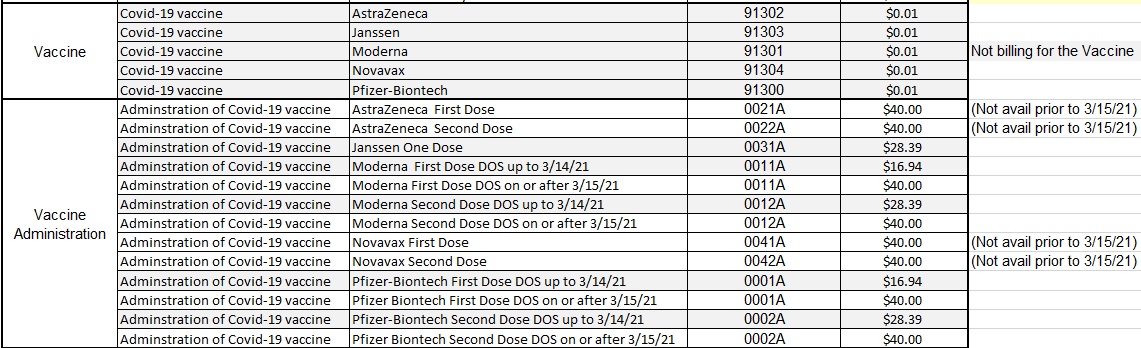
Medicare payment rates for administering doses of approved COVID-19 vaccines.
Reimbursement went up in March 2021, boosting rates from around $16.94-$28.39 up to $40 per dose. Unfortunately, our client has experienced that not all payers are fully reimbursing the $40 for administering the vaccine. Recently, this client asked for help to see if they could balance bill the remainder to the Health Resources and Services Administration (HRSA) because they had heard about government funding that might cover this. I researched the topic and found there is indeed funding available.
Getting paid through the COVID-19 Coverage Assistance Fund
There is a program called HRSA Covid-19 Coverage Assistance Fund. This fund was created to cover the cost of administering COVID-19 vaccines to patients whose health insurance does not cover, or “fully” cover the vaccine. Providers and Mass Immunizers, like this lab, who have administered FDA authorized vaccines can submit claims for consideration of coverage assistance. To be eligible for the reimbursement, they must first have submitted the claim to the individual health plan, and the claim must have been either denied or partially paid.
Section 3203 of the CARES Act requires insurers to cover any qualifying coronavirus preventative service, including COVID-19 vaccines and their administration. The Act also states that this coverage should be applied without imposing any cost-sharing requirements, such as a copayment, co-insurance, or deductible.
Currently, those who administered the vaccine must administer it regardless of the recipient’s ability to pay. Providers are not allowed to seek any reimbursement from recipients, but they are allowed to seek reimbursement from insurance plans that cover it. Vaccine administration fees for the uninsured are covered through HRSA’s Uninsured program.
Items to remember when billing the fund
Providers and Mass Immunizers, like this lab, can request claims reimbursement though the COVID-19 Coverage Assistance Fund electronically and will be reimbursed at the national Medicare rate. This includes reimbursement for patient charges related to COVID-19 vaccination such as copayments, deductibles, and co-insurance. There is also no requirement to ascertain a patient’s immigration status in order to receive reimbursement from the fund.
However, providers are required to first verify that there is no other payer available to bill who will reimburse them the underpaid amount. They also have to attest that they will accept the defined program reimbursement as payment in full and agree to not balance billing the patient.
For COVID-19 vaccinations administered in-home* with dates of service on or after June 8, 2021, reimburses at a rate of $75.50 per administered dose. Effective August 12, 2021. This includes an additional third dose for certain immunocompromised individuals.
*For a definition of in-home administration click here (PDF).
If you have questions about becoming a Mass Immunizer, or need assistance navigating reimbursement through the Coverage Assistance Fund, please feel free to contact us.
Is there any assistance for COVID testing that is denied by insurance?
Hi, Lisa. I followed up with the author of this article, and she confirmed there currently is no additional assistance for denied testing claims outside of appealing the denial with proper documentation. However, HRSA covering denied/underpaid testing claims is currently a proposal that is being discussed. Hope this helps!